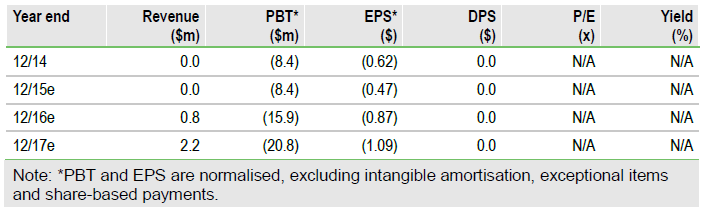
New pancreatic data and a CE mark
VolitionRx (N:VNRX) recently had data published in the peer-reviewed journal Clinical Epigenetics, which confirmed the utility of the NuQ platform for detecting pancreatic cancer. Also, combining NuQ assays with the CA 19-9 biomarker, it was able to achieve 92% sensitivity and 90% specificity. Additionally, the company announced interim data from a 4,800-subject trial in colorectal cancer (CRC) and received its first CE mark in Europe for an assay for the detection of CRC.
Promising pancreatic data published
Full results from a 59-subject trial for detecting pancreatic cancer in blood serum using the VolitionRx NuQ platform were published in the journal Clinical Epigenetics. A panel of five NuQ assays achieved 84% sensitivity and 90% specificity in differentiating subjects with cancer from healthy ones. Importantly, combining four NuQ assays with CA 19-9, a biomarker for pancreatic cancer that is also measured in blood serum, achieved 92% sensitivity and 90% specificity.
To Read the Entire Report Please Click on the pdf File Below